Food Commercial Sterility Testing and Equipment
Item No.: YTEST-2-4157
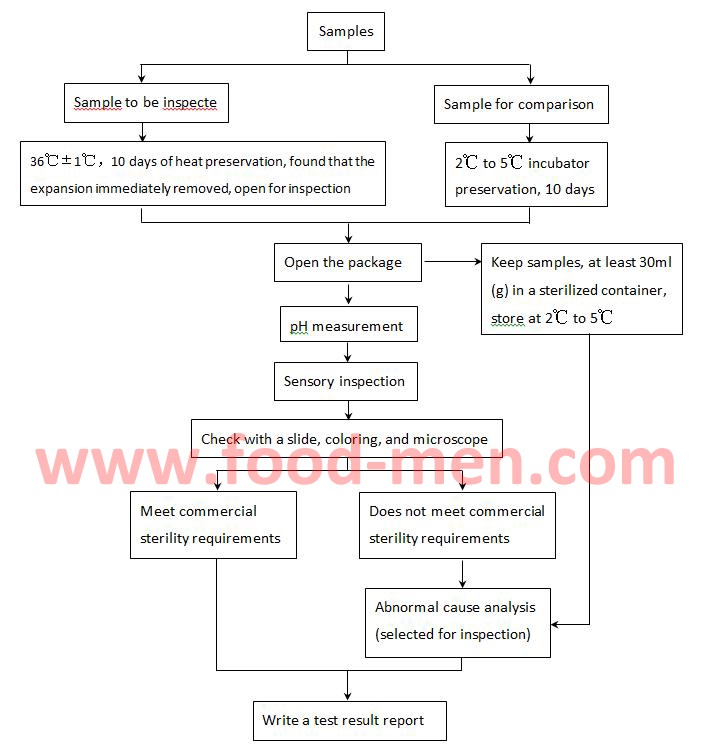
Commercial sterility tests are essential for food of cans. This article introduces methods and equipment for commercial sterility testing to meet FDA requirements.
| Product parameters |
| Product Name: |
Food Commercial Sterility Testing Equipment |
| Model Number: |
TEST-1 |
| Brand: |
FOOD EYES |
| Minimum Order Quantity: |
1 single equipment or instrument for food commercial sterility testing. |
| Price: |
The price is determined according to the parameters and quantity of the order. |
| Supply Ability: |
15 sets (complete sets) of food commercial sterility testing equipment per month. |
| Place of Origin: |
China |
INQUIRY